Lithium-metal batteries could exhibit significantly higher energy densities than lithium-ion batteries, which are the primary battery technology on the market today. Yet lithium-metal cells also typically have significant limitations, the most notable of which is a short lifespan.
Researchers at University of Science and Technology of China and other institutes recently introduced a new electrolyte design that could be used to develop highly performing lithium-metal pouch cells with longer lifespans. This electrolyte, presented in a paper in Nature Energy, has a unique nanometer-scale solvation structure, with pairs of ions densely packed together into compact ion-pair aggregates (CIPA).
"The primary objectives of our recent work are to markedly accelerate the practical applications of lithium-metal batteries and offer deep mechanistic understandings of this complicated system," Prof. Shuhong Jiao, co-author of the paper, told Tech Xplore.
"Li-metal batteries are the holy grail of the battery field and viewed as a promising next-generation battery technique, because they have ultra-high energy density, theoretically >500 Wh/kg. This is greater than 2-fold compared with today's lithium-ion batteries dominating the battery market, which means that if we can replace lithium-ion batteries with lithium-metal batteries, the range of electric vehicles can be doubled per charge."
Lithium-metal batteries introduced to date have a highly limited cycle life of approximately 50 cycles, which is significantly lower than that of commercial lithium-ion batteries, which can usually retain their good performance for approximately 1,000 cycles. The reasons behind this lower lifespan are the growth of lithium dendrites, the high reactivity of lithium-metal and high-voltage transition metal cathodes, which collectively prompt the constant degradation of the electrolyte.
"Despite the extensive efforts of researchers around the world, the performance of lithium-metal batteries is still far from satisfactory (>500 Wh/kg, 1,000 cycles)," Prof. Jiao said. "The primary cause is that the interfaces between electrolyte and electrodes (i.e., the anode-electrolyte interface and cathode-electrolyte interface) cannot be completely stabilized as in the case of lithium-ion batteries. Constant and severe electrolyte degradation still occurs during the battery operation."
Approximately five years ago, Prof. Jiao and her colleagues designed an electrolyte that can simultaneously stabilize anode-electrolyte and cathode-electrolyte interfaces in lithium-metal battery cells, suppressing the electrolyte's degradation. Their electrolyte design builds on early investigations of microscopic physicochemical processes inside lithium-metal batteries.
"An electrolyte is a key component of lithium-metal batteries, since it can tune the chemistry/structure of SEI and thus guide the plating behavior of lithium-metal, eventually dictating the battery performance," Prof. Jiao explained.
"For practical application purposes, we tried to realize this by using cheap components. The countless works of other researchers in this field also inspired us a lot, as they introduced many new classes of electrolyte like highly concentrated electrolyte, localized high-concentration electrolyte, weak-solvating electrolyte and liquefied-gas electrolyte, etc."
To carry out this recent study, Prof. Jiao and her research group teamed up with other teams that could perform theoretical calculations and could characterize electrolytes at a microscopic scale. Their collaborative efforts ultimately led to the design of a new class of electrolytes that can extend the lifespan of lithium-metal batteries.
The electrolytes they designed are made of commercially available and affordable molecules. Their characterizing feature is their unique solvation structure.
"Solvation structure is a crucial inherent feature of an electrolyte, since it governs the interfacial behavior of electrolyte, like its interfacial reaction mechanism that controls the formation of SEI and thus the SEI chemistry and structure," Prof. Jiao said.
"The electrolyte solvation structure has been intensively tailored at the microscopic level in the scientific peer-reviewed literature so far, particularly the lithium ion's first solvation shell, but the structural tuning beyond this scale, namely the second solvation shell and beyond, is largely overlooked."
The recent study by Prof. Jiao and her colleagues has pioneered the tuning of an electrolyte's solvation structure at the mesoscopic level. Their unique design specifically focuses on the interaction between ion pairs underlying the formation of the electrolyte's aggregate structure.
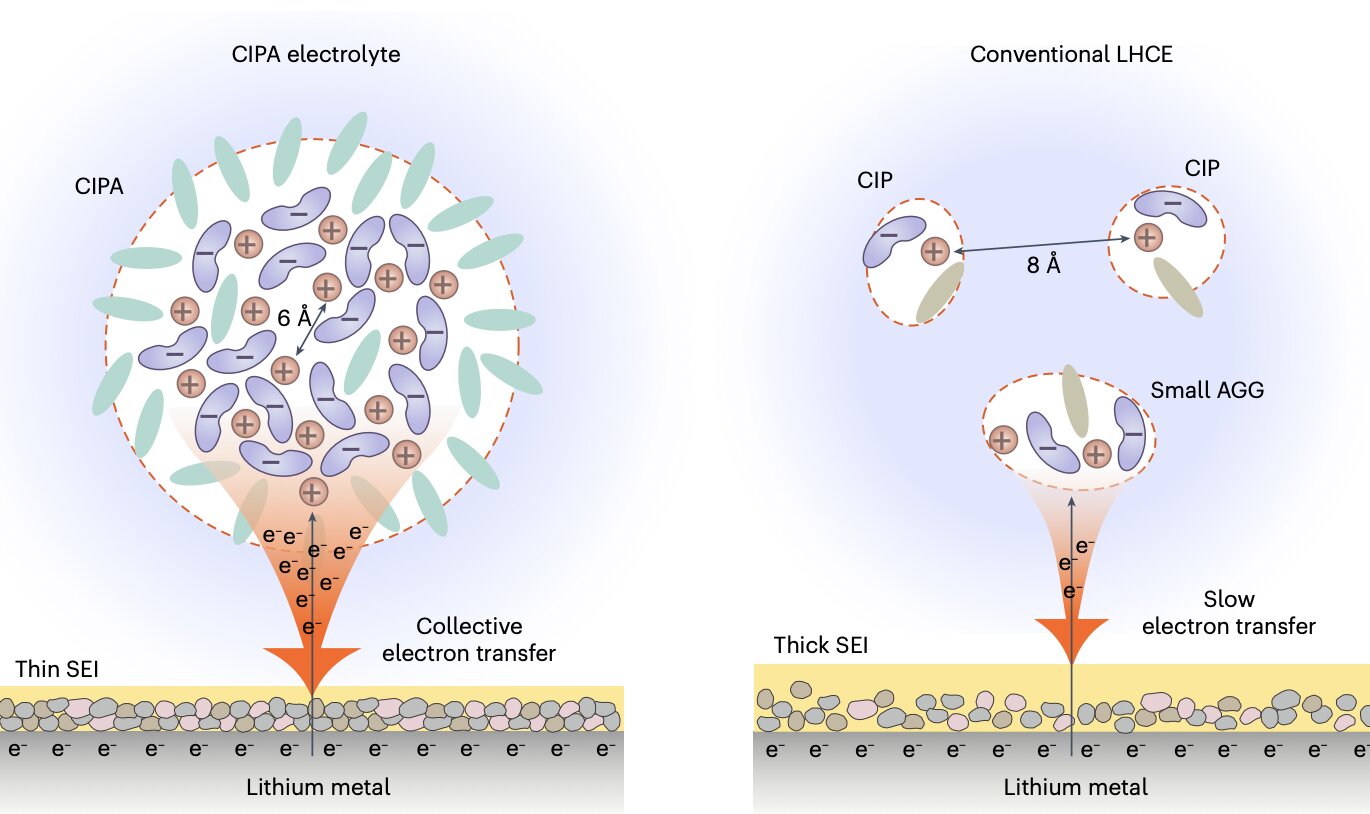
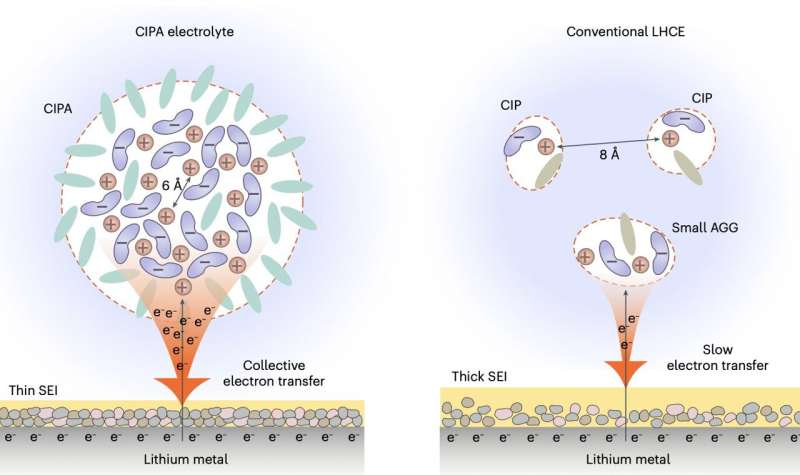
"Our electrolyte features large compact aggregates, which are formed by the dense packing of lithium-anion ion pairs with coordination bonding between each other, which we define as the 'compact ion-pair aggregate (CIPA)," Prof. Jiao said. "This marks stark contrast to the domination of small aggregates and separate ion pairs in the localized high-concentration electrolyte, a state-of-the-art electrolyte class with leading battery performance to-date, opening a new avenue for electrolyte design."
Notably, the new electrolyte designed by this research team exhibits a unique collective reduction on the lithium-metal anode. This means that clouds of anions in the CIPA structure are rapidly reduced (i.e., decomposed) on the surface of the lithium, forming inorganic compounds such as Li2O and LiF, as well as a thin and stable SEI, which in turn suppresses the constant decomposition of the electrolyte.
"Thanks to the unique collective electron transfer behavior, our electrolyte forms a thin and conformal SEI with low organic content and rich in inorganic components with uniform distribution, which can promote the homogeneous lithium ion flux inside the SEI and dendrite-free lithium deposition," Prof. Jiao said. "This leads to a homogeneous and compact lithium deposition, which decreases the specific areas of lithium-metal anode to further suppress the electrolyte decomposition."
Moreover, the researchers' newly designed electrolyte simultaneously exhibits good oxidative stability and suppresses the dissolution of transition metal elements from the cathode, thus improving the stability of the cathode interface. The stabilization of this interface, along with that of the lithium-electrolyte-interface, was found to translate to stable cycling for a prolonged number of cycles.
"The mesoscopic solvation structure introduced in our paper leads to a new class of electrolytes, opening up a new avenue for electrolyte design of lithium-metal batteries," Prof. Jiao said.
To assess the potential of their newly designed electrolyte, the researchers used it to create a 500 Wh/kg lithium-metal pouch cell. In initial tests, this cell was found to retain 91% of its energy after operating for 130 cycles. In the future, this new electrolyte design could be reproduced and tested by other researchers worldwide, to further assess its potential for extending the life of lithium-metal batteries.
"We are now planning to further prolong the cycle life of 500 Wh/kg lithium-metal pouch cells to more than 1,000 cycles," Prof. Jiao added. "On the other hand, we are still exploring the new battery system to realize much higher energy density with a long lifespan, such as ≥ 600 Wh/kg with 100-200 cycles. All these fundamental scientific research studies are valuable to realize the deployment of lithium-metal batteries in many scenes."
More information: Yulin Jie et al, Towards long-life 500 Wh kg−1 lithium metal pouch cells via compact ion-pair aggregate electrolytes, Nature Energy (2024). DOI: 10.1038/s41560-024-01565-z
© 2024 Science X Network
Citation: Novel electrolyte design shows promise for longer-lasting lithium-metal batteries (2024, August 18) retrieved 18 August 2024 from https://techxplore.com/news/2024-08-electrolyte-longer-lithium-metal-batteries.html
This document is subject to copyright. Apart from any fair dealing for the purpose of private study or research, no part may be reproduced without the written permission. The content is provided for information purposes only.