by Joseph E. Harmon,
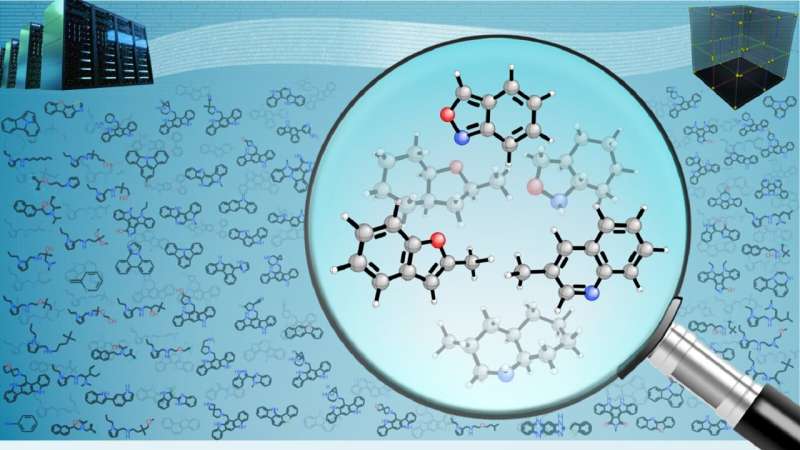
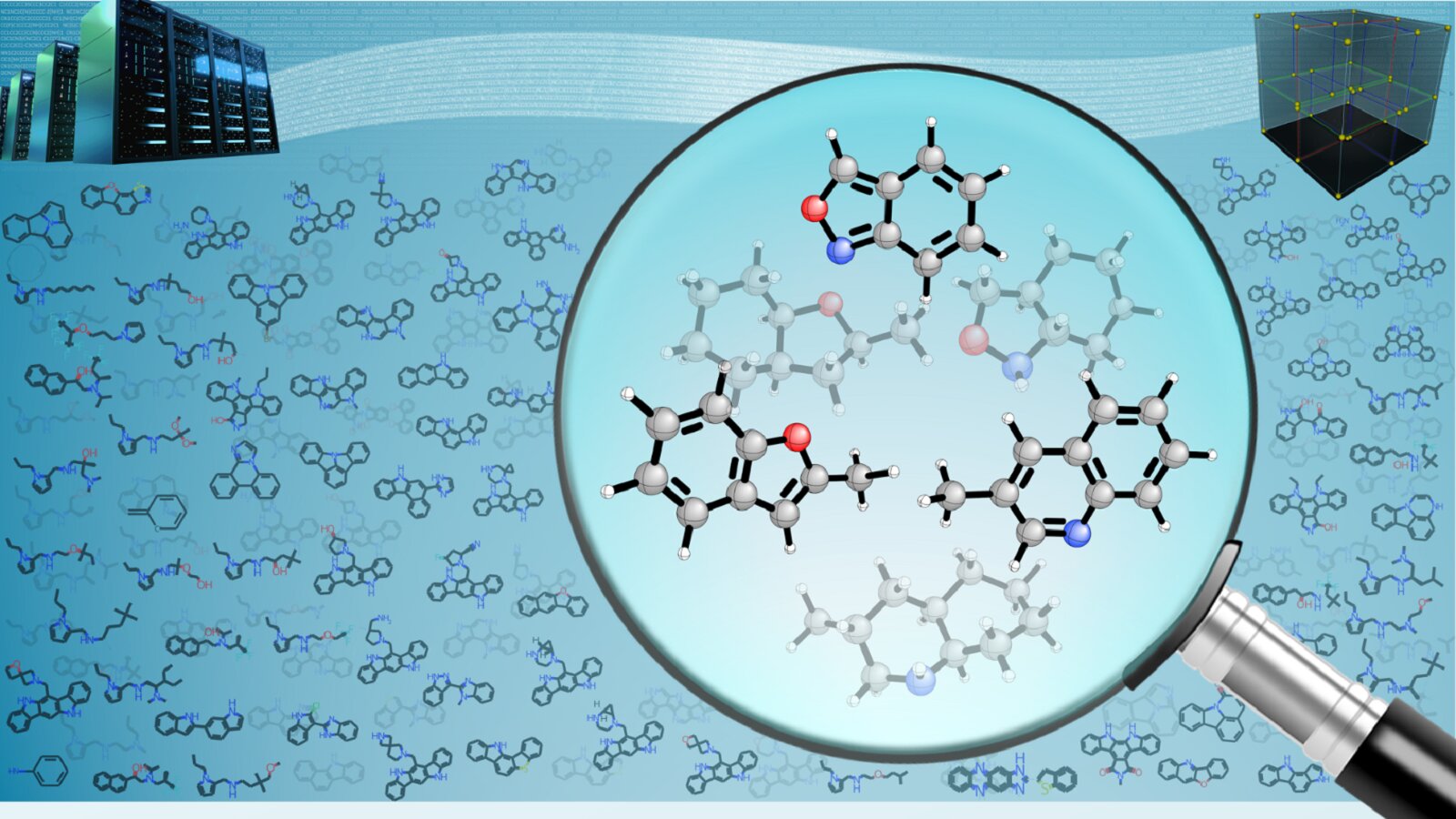
In a computational study leveraging artificial intelligence (AI), scientists at the U.S. Department of Energy's (DOE) Argonne National Laboratory have assessed 160 billion molecules, a number exceeding the people born in the entire span of human history. Their goal was to screen the molecules for suitability as liquid carriers of hydrogen.
The findings are published in the journal Digital Discovery.
The sun is essentially an enormous ball of mostly hydrogen gas, releasing energy in the form that warms the Earth and the rest of our solar system. Because of its energy content and abundance, hydrogen shows great promise as an energy source on Earth as well. It could be fueling cars, trucks, buses, trains and ships and generating electricity for consumers. While the sun's energy comes from the fusion of hydrogen atoms, the process the team aims to use on Earth involves the combustion of hydrogen.
Hydrogen in its pure form exists as a gas under normal conditions. For use as a fuel, one of the challenges is shipping this gas safely to refueling stations and storing it. Hydrogen carrier compounds in liquid form, however, have several advantages. They have a much better safety profile because they are not as prone to leaking and explosion. They also have a much higher energy content per unit volume, making storage and transportation far easier.
"The liquid compound form would essentially eliminate certain problems with pure hydrogen gas, especially given that there is a well-established infrastructure in place for storing and transporting gasoline and other liquid chemicals safely," said Rajeev Surendran Assary, a chemist and Argonne group leader in the Materials Science division.
The most visible form of a liquid hydrogen carrier compound is water—two atoms of hydrogen and one of oxygen. Another form is organic molecules, essentially an endless number of possible combinations of hydrogen and carbon atoms, in addition to other atoms such as nitrogen and oxygen.
"Assisted by AI, we are searching for organic liquid molecules that, through a low-cost chemical reaction with a catalyst, one could alternately add or release hydrogen for use as a fuel," said computational scientist Logan Ward in the Data Science and Learning division. Of key importance would be that this reaction does not add carbon to the atmosphere. That is, it must be carbon neutral.
"We were looking for organic liquid molecules that hold on to hydrogen for a long time, but not so strongly that they could not be easily removed on demand," said postdoctoral appointee Hassan Harb in the Materials Science division. "They must also have the capacity to store sufficient hydrogen for practical use." After the hydrogen removal, replacement hydrogen would be added to the liquid for reuse.
Among the billions of possible liquid hydrogen carriers, common examples include chemicals like ammonia and methanol. However, the relatively few candidates tested in the laboratory to date have suffered from chemical instability and unwanted side reactions.
The team screened the candidate molecules based on four factors. One was structural similarity to known liquid hydrogen carriers. Another was desirable physical properties, such as melting and boiling points—the liquid must stay liquid when the hydrogen has been added or extracted. Third is that the liquid must be able to store a large amount of hydrogen per unit volume. Finally, the amount of energy needed to release the hydrogen from the liquid must be low enough.
"We started by accessing chemical databases with data on organic molecules," said postdoctoral appointee Sarah Elliott in the Chemical Sciences and Engineering division. "We found over 160 billion such molecules, and combining AI with the latest theoretical computational methods is essential to screen this enormous army of molecules for the best ones."
The team's calculations necessitated access to supercomputers available at few places in the world. One of them is Argonne, home to the Argonne Leadership Computing Facility, a DOE Office of Science user facility. The team also relied on Bebop, a computing cluster operated by the Laboratory Computing Resource Center at Argonne.
Even with these powerful resources available, if one allots one millisecond of computing time per molecule, that translates into five years of computing time for 160 billion molecules. For that reason, the team developed an AI-based screening approach that sped up the computations to three million molecules per second, or about 14 hours for the 160 billion.
"This turns the computations from something we could do only once in a whole project, if at all, to something we can do overnight and repeat as we get feedback from computation and experiment," Ward said.
By their unique approach, the team whittled down the candidates from over 160 billion to a mere 41. Now, the task passes into the hands of experimentalists to test the promising ones. The team's computational approach paves the way for a new era of innovation in sustainable energy solutions.
In addition to Assary, Elliott, Ward and Harb, authors include Ian Foster, Stephen Klippenstein and Larry Curtiss.
More information: Hassan Harb et al, Uncovering novel liquid organic hydrogen carriers: a systematic exploration of chemical compound space using cheminformatics and quantum chemical methods, Digital Discovery (2023). DOI: 10.1039/D3DD00123G
Citation: AI helps whittle down candidates for hydrogen carriers in liquid form from billions to about 40 (2024, January 10) retrieved 10 January 2024 from https://techxplore.com/news/2024-01-ai-whittle-candidates-hydrogen-carriers.html
This document is subject to copyright. Apart from any fair dealing for the purpose of private study or research, no part may be reproduced without the written permission. The content is provided for information purposes only.